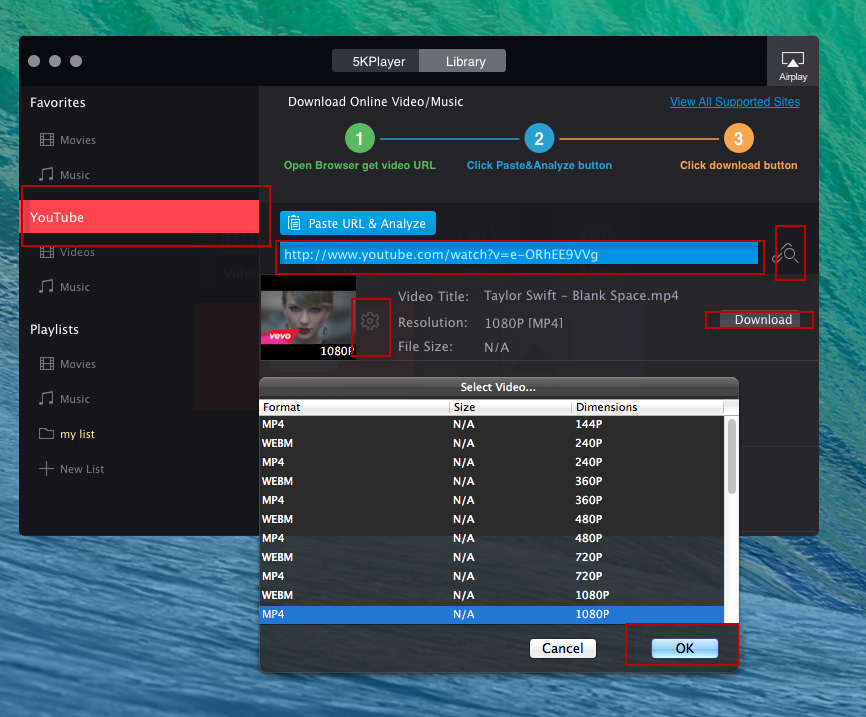
Maier B, Leader AM, Chen ST, Tung N, Chang C, LeBerichel J, Chudnovskiy A, Maskey S, Walker L, Finnigan JP, Kirkling ME, Reizis B, Ghosh S, D’Amore NR, Bhardwaj N, Rothlin CV, Wolf A, Flores R, Marron T, Rahman AH, Kenigsberg E, Brown BD, Merad M (2020) A conserved dendritic-cell regulatory program limits antitumour immunity. Zilionis R, Engblom C, Pfirschke C, Savova V, Zemmour D, Saatcioglu HD, Krishnan I, Maroni G, Meyerovitz CV, Kerwin CM, Choi S, Richards WG, De Rienzo A, Tenen DG, Bueno R, Levantini E, Pittet MJ, Klein AM (2019) Single-cell transcriptomics of human and mouse lung cancers reveals conserved myeloid populations across individuals and species. Īrdouin L, Luche H, Chelbi R, Carpentier S, Shawket A, Montanana Sanchis F, Santa Maria C, Grenot P, Alexandre Y, Gregoire C, Fries A, Vu Manh TP, Tamoutounour S, Crozat K, Tomasello E, Jorquera A, Fossum E, Bogen B, Azukizawa H, Bajenoff M, Henri S, Dalod M, Malissen B (2016) Broad and largely concordant molecular changes characterize tolerogenic and immunogenic dendritic cell maturation in thymus and periphery. Mattiuz R, Brousse C, Ambrosini M, Cancel JC, Bessou G, Mussard J, Sanlaville A, Caux C, Bendriss-Vermare N, Valladeau-Guilemond J, Dalod M, Crozat K (2021) Type 1 conventional dendritic cells and interferons are required for spontaneous CD4(+) and CD8(+) T-cell protective responses to breast cancer. Īlexandre YO, Cocita CD, Ghilas S, Dalod M (2014) Deciphering the role of DC subsets in MCMV infection to better understand immune protection against viral infections. Vu Manh TP, Bertho N, Hosmalin A, Schwartz-Cornil I, Dalod M (2015) Investigating evolutionary conservation of dendritic cell subset identity and functions. We hope that this method will be helpful for both wet lab and bioinformatics researchers interested in harnessing scRNAseq data for deciphering the biology of DCs or other cell types and that it will contribute to establishing high standards in the field. It is accompanied with a more complete tutorial on GitHub. We describe this pipeline step-by-step, including data quality controls, dimensionality reduction, cell clustering, cell cluster annotation, inference of the cell activation trajectories, and investigation of the underpinning molecular regulation. 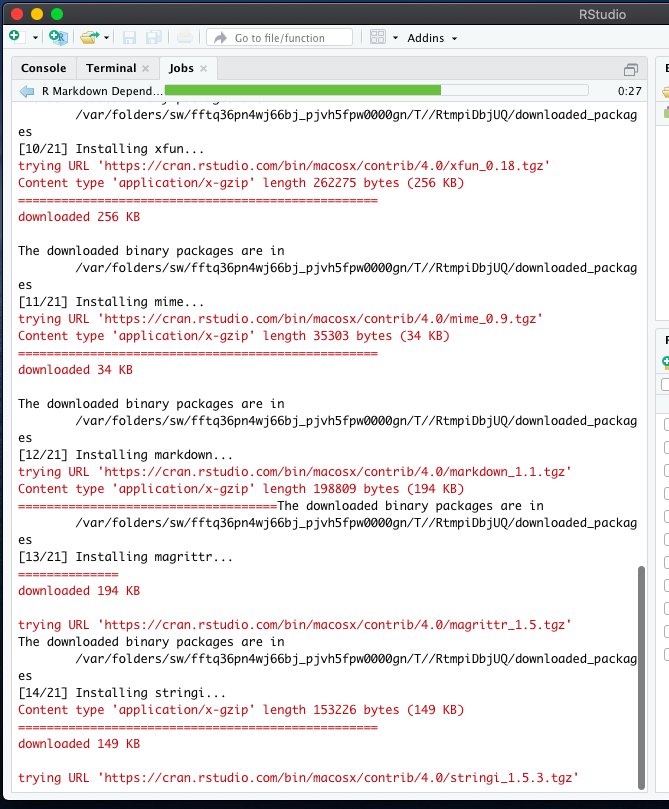
In this chapter, we take these issues into account for providing a pipeline for scRNAseq analysis and illustrating it with a tutorial reanalyzing a public dataset of mononuclear phagocytes isolated from the lungs of naïve or tumor-bearing mice. It is also important to emphasize the necessity of examining whether similar cell activation trajectories are inferred by using different, complementary methods. In addition, awareness must be raised on the need for specific, robust, and tractable strategies to annotate cells for cell type identity and activation states. However, for new users of this approach, determining which analytics strategy and computational tools to choose can be quite challenging, considering the rapid evolution and broad burgeoning in the field. 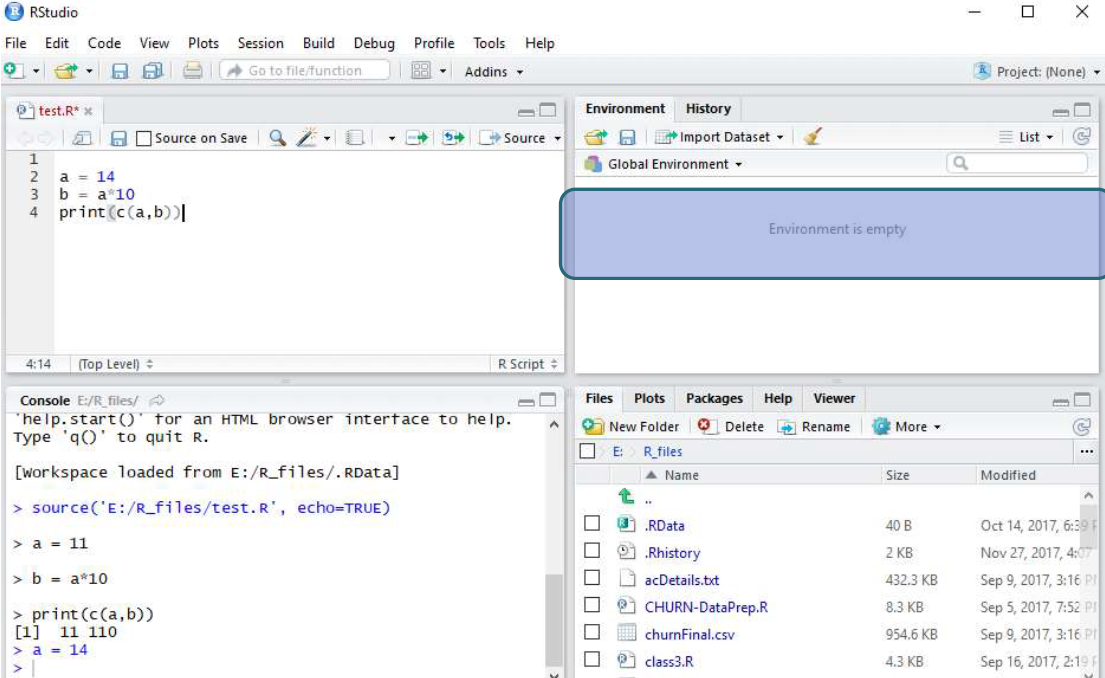
To decipher the nature, functions, and regulation of DC types and their physiological activation states, one of the methods that can be harnessed most successfully is ex vivo single-cell RNA sequencing (scRNAseq). 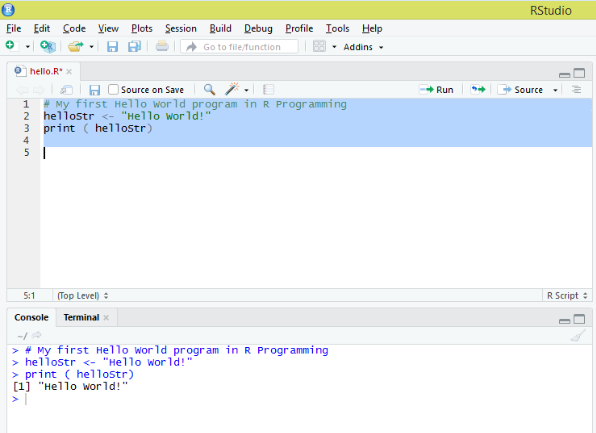
Hence, to better understand DC biology and harness it in the clinic, we must determine which combinations of DC types and activation states mediate which functions and how. Second, each DC type can undergo different activation states, fine-tuning its functions depending on its tissue microenvironment and the pathophysiological context, by adapting the output signals it delivers to the input signals it receives. First, DCs encompass distinct cell types specialized in different functions. Hence, DCs are very plastic, which results from two key characteristics. Dendritic cells (DCs) orchestrate innate and adaptive immunity, by translating the sensing of distinct danger signals into the induction of different effector lymphocyte responses, to induce the defense mechanisms the best suited to face the threat.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
